Atopic Dermatitis
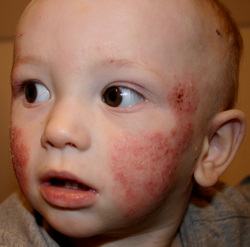
Atopic dermatitis is a chronic inflammatory skin condition that commonly presents in early childhood. 80% of cases develop during the first year of life and 90% develop before age 5. Atopic dermatitis is part of an atopic syndrome which also includes allergic rhinitis/conjunctivitis, food allergies and allergic bronchial asthma. There is a strong genetic link to atopic dermatitis, with a risk of 70% if both parents also had atopic dermatitis. Other risk factors include higher social class families and children from small families. Higher levels of IgE are also
associated with increased incidence of atopic dermatitis as well as severity of atopic dermatitis.
Probiotics have been suggested as both a prevention and treatment option for atopic dermatitis early in life. One of the main proposed mechanisms of action of probiotics in the prevention of atopic dermatitis is related to the hygiene hypothesis. The hygiene hypothesis links a decrease in exposure to microbes early in life with a decreased immune system and an increased susceptibility to disorders like allergies. Changes in microbial flora in infancy are also thought to play a roll, which is where probiotics are postulated to have their benefit.
Evidence for Prevention:
Several meta-analyses have looked at probiotics and the prevention of atopic dermatitis and IgE associated atopic dermatitis in
young children. Many different probiotic strains have been researched and some studies looked at probiotic use during pregnancy
while others looked at probiotic use in infancy. Overall there seems to be a reduction in atopic dermatitis in children associated with
probiotic use. Consider probiotics containing Lactobacillus rhamnosus GG for prevention. Culturelle is an example of a product
containing L rhamnosus GG.
Meta-Analysis 1 – Probiotic supplementation during pregnancy or infancy for the prevention of atopic dermatitis
-the meta-analysis was conducted in Italy in 2011, published in 2012
Trial Components2:
· included 18 papers with results on probiotics in the prevention of atopic dermatitis, based on 14 different trials
· all were randomized, placebo-controlled trials
· all were double-blinded
· 2 studies looks at the use of probiotics and prebiotics together
· 11 studies considered severity of atopic dermatitis
Results2:
· there was a decreased incidence of atopic dermatitis with probiotic use vs. placebo with a RR= 0.79 (95% CI=0.71-0.88)
· there was a decreased incidence of IgE-associated atopic dermatitis with probiotic use vs. placebo with a RR= 0.80 (95% CI=0.66-0.96)
· the effect was similar regardless of the time of probiotic use (pregnancy, infancy, or both)
· of the 11 studies that considered severity of atopic dermatitis, 9 reported no difference between the treatment and placebo groups
· risk estimates with use of >1 type of probiotic were slightly lower than those for interventions based on a single probiotic type
· probiotic strains containing L. rhamnosus GG were included in 6 trials and showed a RR of 0.74 (95% CI=0.61-0.90)
Evidence for Treatment:
At this time there is inconclusive evidence to recommend probiotics in the treatment of atopic dermatitis in children. There have been
some studies to support probiotic use, but the majority of studies and meta-analyses suggest that probiotics taken for treatment do not
statistically reduce atopic dermatitis in children.
Cochrane Review3:
· assessed 12 randomised controlled trials
· involved a total of 781 participants, all children
· found no significant difference in symptom severity after treatment with probiotics; severity was measured on a scale from 0 to 20 and was 0.90 points lower after probiotic treatment compared to placebo (95% CI -1.04, 2.84; p=0.36)
· there was no significant difference in participant or parent-rated eczema severity
· there was no significant difference in investigator rated severity
· authors concluded “the evidence suggests that probiotics are not an effective treatment for eczema...”
Trial 1- Treatment and secondary prevention effects of the probiotics Lactobacillus paracasei or Bifodobacterium lactis on early infant
eczema: randomized controlled trial with follow-up until age 3 years
Study Components4:
· randomized, placebo-controlled trial & an observational arm
· 137 infants age 3-6 months were randomized to either Lactobacillus paracasei CNCM I-2116 1010 colony-forming units, Bifidobacterium lactis CNCM I-3446 1010 colony-forming units or placebo (Maltodextrin) for a period of 3 months
· follow up was conducted for 3 years
· all infants were put on a dairy elimination diet (hydrolyzed whey-formula) in the randomization arm
· observational arm looked at exclusively breast-fed infants and infants on standard formula
· standardized skin-treatment of 1% hydrocortisone ointment 2x/day, emollients/moisturizers 2-4x/day and bath emollient were used
· Primary outcome: SCORAD index was calculated after a 12-week intervention
· Secondary outcome: SCORAD index was calculated that included: IDQoL-score (infant dermatitis quality of life), gastrointestinal permeability, urinary eosinophilic protein X (U-EPX), presence/absence of study strains in stools, prevalence of sensitization, symptoms of allergic disease at age 12, 18 and 36 months
Results4:
· SCORAD score decreased significantly over time, but there was no significant difference in the reduction between the three groups
· when allergic sensitization was controlled or when only sensitized infants were included, there was still no significant difference
· no significant differences were seen in the observational arm
· no significant differences were seen in any of the secondary outcomes
Appraisal and Comments4:
Positive aspects of this trial include randomization, specifics about which strain and quantity of probiotics were used, large study size and long follow-up. After this study was designed information suggested that probiotics may only work in sensitized patients. Although this was not originally controlled for in this study, retrospective analysis suggested that outcomes remained the same when only sensitized infants were observed. A study that includes sensitization in their design is needed to further evaluate this.
Prebiotics for the Treatment of Atopic Dermatitis5:
There are very few randomized controlled trials assessing the benefits of prebiotics in the treatment of atopic dermatitis and other allergic diseases. One small randomized, double-blind study of 29 infants and children <3 years showed benefit in treatment with the prebiotic kestose, a fructo-fructooligosaccharide, versus placebo (maltose) over a 12 week period. Results were a significant reduction in median SCORAD (Severity Scoring of Atopic Dermatitis) scores between the kestose and placebo groups at week 6 (25.3 vs. 36.4; P=0.004) and week 12 (19.5 vs. 37.5; P<0.001).
Further studies are needed to confirm the benefits of prebiotics in the treatment of atopic dermatitis.
1) Sibbald D. Atopic, Contact, and Stasis Dermatitis. In: Patient Self-Care. 2nd ed. Ottawa, ON: Canadian Pharmacists Association, (2010): 510/520.
2) Pelucchi C. et. al. Probiotics supplementation during pregnancy or infancy for the prevention of atopic dermatitis- a meta-analysis. Epidemiology. 23 (2012): 402-414.
3) Boyle R, Bath-Hextall F, Leonardi-Bee J, Murrell DF, Tang M. Probiotics for treating eczema. Allergy and Clincal Immunology 2008; 4.
4) Gore C. et. al. Treatment and secondary prevention effects of the probiotics Lactobacillus paracasei or Bifodobacterium lactis on early infant
eczema: randomized controlled trial with follow-up until age 3 years. Clinical & Experimental Allergy. 42 (2011): 112-122.
5) Shibata R. Clinical effects of kestose, a prebiotic oligosaccharide, on the treatment of atopic dermatitis in infants. Clinical & Experimental Allergy. 39.9 (2009): 1397-1403.
associated with increased incidence of atopic dermatitis as well as severity of atopic dermatitis.
Probiotics have been suggested as both a prevention and treatment option for atopic dermatitis early in life. One of the main proposed mechanisms of action of probiotics in the prevention of atopic dermatitis is related to the hygiene hypothesis. The hygiene hypothesis links a decrease in exposure to microbes early in life with a decreased immune system and an increased susceptibility to disorders like allergies. Changes in microbial flora in infancy are also thought to play a roll, which is where probiotics are postulated to have their benefit.
Evidence for Prevention:
Several meta-analyses have looked at probiotics and the prevention of atopic dermatitis and IgE associated atopic dermatitis in
young children. Many different probiotic strains have been researched and some studies looked at probiotic use during pregnancy
while others looked at probiotic use in infancy. Overall there seems to be a reduction in atopic dermatitis in children associated with
probiotic use. Consider probiotics containing Lactobacillus rhamnosus GG for prevention. Culturelle is an example of a product
containing L rhamnosus GG.
Meta-Analysis 1 – Probiotic supplementation during pregnancy or infancy for the prevention of atopic dermatitis
-the meta-analysis was conducted in Italy in 2011, published in 2012
Trial Components2:
· included 18 papers with results on probiotics in the prevention of atopic dermatitis, based on 14 different trials
· all were randomized, placebo-controlled trials
· all were double-blinded
· 2 studies looks at the use of probiotics and prebiotics together
· 11 studies considered severity of atopic dermatitis
Results2:
· there was a decreased incidence of atopic dermatitis with probiotic use vs. placebo with a RR= 0.79 (95% CI=0.71-0.88)
· there was a decreased incidence of IgE-associated atopic dermatitis with probiotic use vs. placebo with a RR= 0.80 (95% CI=0.66-0.96)
· the effect was similar regardless of the time of probiotic use (pregnancy, infancy, or both)
· of the 11 studies that considered severity of atopic dermatitis, 9 reported no difference between the treatment and placebo groups
· risk estimates with use of >1 type of probiotic were slightly lower than those for interventions based on a single probiotic type
· probiotic strains containing L. rhamnosus GG were included in 6 trials and showed a RR of 0.74 (95% CI=0.61-0.90)
Evidence for Treatment:
At this time there is inconclusive evidence to recommend probiotics in the treatment of atopic dermatitis in children. There have been
some studies to support probiotic use, but the majority of studies and meta-analyses suggest that probiotics taken for treatment do not
statistically reduce atopic dermatitis in children.
Cochrane Review3:
· assessed 12 randomised controlled trials
· involved a total of 781 participants, all children
· found no significant difference in symptom severity after treatment with probiotics; severity was measured on a scale from 0 to 20 and was 0.90 points lower after probiotic treatment compared to placebo (95% CI -1.04, 2.84; p=0.36)
· there was no significant difference in participant or parent-rated eczema severity
· there was no significant difference in investigator rated severity
· authors concluded “the evidence suggests that probiotics are not an effective treatment for eczema...”
Trial 1- Treatment and secondary prevention effects of the probiotics Lactobacillus paracasei or Bifodobacterium lactis on early infant
eczema: randomized controlled trial with follow-up until age 3 years
Study Components4:
· randomized, placebo-controlled trial & an observational arm
· 137 infants age 3-6 months were randomized to either Lactobacillus paracasei CNCM I-2116 1010 colony-forming units, Bifidobacterium lactis CNCM I-3446 1010 colony-forming units or placebo (Maltodextrin) for a period of 3 months
· follow up was conducted for 3 years
· all infants were put on a dairy elimination diet (hydrolyzed whey-formula) in the randomization arm
· observational arm looked at exclusively breast-fed infants and infants on standard formula
· standardized skin-treatment of 1% hydrocortisone ointment 2x/day, emollients/moisturizers 2-4x/day and bath emollient were used
· Primary outcome: SCORAD index was calculated after a 12-week intervention
· Secondary outcome: SCORAD index was calculated that included: IDQoL-score (infant dermatitis quality of life), gastrointestinal permeability, urinary eosinophilic protein X (U-EPX), presence/absence of study strains in stools, prevalence of sensitization, symptoms of allergic disease at age 12, 18 and 36 months
Results4:
· SCORAD score decreased significantly over time, but there was no significant difference in the reduction between the three groups
· when allergic sensitization was controlled or when only sensitized infants were included, there was still no significant difference
· no significant differences were seen in the observational arm
· no significant differences were seen in any of the secondary outcomes
Appraisal and Comments4:
Positive aspects of this trial include randomization, specifics about which strain and quantity of probiotics were used, large study size and long follow-up. After this study was designed information suggested that probiotics may only work in sensitized patients. Although this was not originally controlled for in this study, retrospective analysis suggested that outcomes remained the same when only sensitized infants were observed. A study that includes sensitization in their design is needed to further evaluate this.
Prebiotics for the Treatment of Atopic Dermatitis5:
There are very few randomized controlled trials assessing the benefits of prebiotics in the treatment of atopic dermatitis and other allergic diseases. One small randomized, double-blind study of 29 infants and children <3 years showed benefit in treatment with the prebiotic kestose, a fructo-fructooligosaccharide, versus placebo (maltose) over a 12 week period. Results were a significant reduction in median SCORAD (Severity Scoring of Atopic Dermatitis) scores between the kestose and placebo groups at week 6 (25.3 vs. 36.4; P=0.004) and week 12 (19.5 vs. 37.5; P<0.001).
Further studies are needed to confirm the benefits of prebiotics in the treatment of atopic dermatitis.
1) Sibbald D. Atopic, Contact, and Stasis Dermatitis. In: Patient Self-Care. 2nd ed. Ottawa, ON: Canadian Pharmacists Association, (2010): 510/520.
2) Pelucchi C. et. al. Probiotics supplementation during pregnancy or infancy for the prevention of atopic dermatitis- a meta-analysis. Epidemiology. 23 (2012): 402-414.
3) Boyle R, Bath-Hextall F, Leonardi-Bee J, Murrell DF, Tang M. Probiotics for treating eczema. Allergy and Clincal Immunology 2008; 4.
4) Gore C. et. al. Treatment and secondary prevention effects of the probiotics Lactobacillus paracasei or Bifodobacterium lactis on early infant
eczema: randomized controlled trial with follow-up until age 3 years. Clinical & Experimental Allergy. 42 (2011): 112-122.
5) Shibata R. Clinical effects of kestose, a prebiotic oligosaccharide, on the treatment of atopic dermatitis in infants. Clinical & Experimental Allergy. 39.9 (2009): 1397-1403.
